The European Union represents a massive opportunity for Indian organic exports, with the spice market alone valued at over €1.1 billion. For procurement managers, tapping into India’s rich agricultural landscape seems like a strategic masterstroke. Yet, a staggering number of shipments never reach their destination. They are stopped at the border, rejected for non-compliance, turning a potential profit into a costly logistical nightmare.
Why? Because the rulebook has changed. The path to the EU market is no longer a simple handshake agreement; it’s a complex labyrinth of stringent regulations. This guide is your map. We’ll walk you through the new laws, demystify the jargon, and provide a clear framework to ensure your organic Indian spices and grains not only meet but exceed EU standards, achieving zero rejections.

The New Rulebook: Understanding EU Organic Law & Regulation (EU) 2018/848
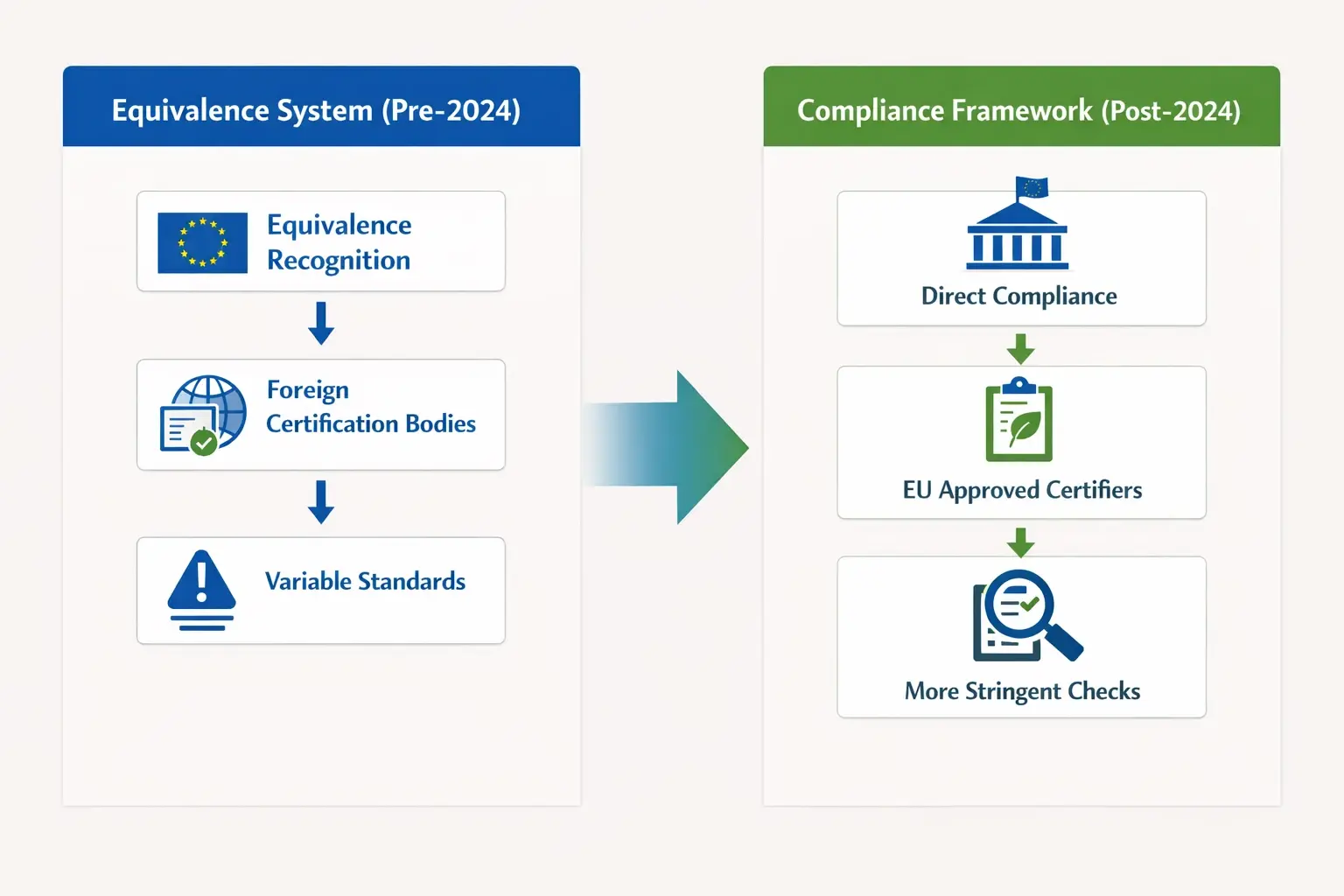
For years, the EU operated on a system of “equivalence.” In simple terms, the EU recognized India’s National Programme for Organic Production (NPOP) as being broadly equivalent to its own standards. If a product was NPOP-certified, it was generally good to go.
That era is over.
As of January 1, 2022, with full enforcement rolling out, the EU has shifted to a much stricter “compliance” framework under Regulation (EU) 2018/848. This isn’t just a minor update; it’s a fundamental change in philosophy.
What’s the difference?
- Equivalence (The Old Way): “Your country’s organic standards are similar enough to ours, so we’ll trust your certification.” This allowed for some flexibility and interpretation.
- Compliance (The New Way): “Your product must meet the exact same stringent rules as a product grown in Germany or France. No exceptions.”
This shift places the full burden of proof on the exporter. It means every single step of the supply chain—from the farm to the processing facility to the final packaging—must adhere to precise EU regulations. There is no more room for “close enough.”
The Organic Paradox: Why NPOP Certification Isn’t Enough
Here is the single biggest “aha moment” for many sourcing managers: achieving an Indian NPOP organic certification is necessary, but it is not sufficient for EU market access. This is the “Organic Paradox.” Many exporters invest heavily in organic certification only to face rejection because they overlooked other, equally critical, layers of EU food safety law.
Think of it as passing your driver’s theory test. It’s an essential first step, but it doesn’t mean you can ignore speed limits or traffic lights.
Successfully entering the EU market requires mastering three distinct pillars of compliance. A failure in any one of them can kill the entire shipment.
The Three Pillars of EU Market Access
Pillar 1: Mastering Organic Certification
Under the new compliance model, your product must be certified by an Indian Control Body (CB) that is officially recognized by the European Commission to certify products in accordance with EU standards. This is a critical distinction. The CB must demonstrate that its auditing and certification for EU-bound products follow the EU rulebook to the letter. Thorough supplier qualification now includes verifying that your partners’ certifiers are on the EU’s approved list for the specific product category you’re sourcing.
Pillar 2: Defeating MRLs – The Silent Killer of Shipments
Maximum Residue Limits (MRLs) are the microscopic gatekeepers of the EU market. Governed by Regulation (EC) No 396/2005, MRLs dictate the highest level of a pesticide residue legally tolerated in or on food. For many substances, the EU’s default MRL is the limit of detection—effectively zero (e.g., 0.01 parts per million).
This is where many “organic” shipments fail. Contamination can happen unintentionally through:
- Pesticide Drift: Wind carrying pesticides from a neighboring non-organic farm.
- Contaminated Soil or Water: Lingering residues from past agricultural practices.
- Cross-Contamination: Occurring during transport or storage.
One of the most notorious culprits is Ethylene Oxide (ETO), a sterilizing agent banned for food use in the EU but sometimes used in other regions. Even trace amounts will trigger an immediate rejection. A robust quality assurance program must include pre-shipment lab testing from NABL-accredited labs to screen for a wide panel of pesticides and contaminants.
Pillar 3: Complying with REACH & Hygiene Standards
Beyond pesticides, the EU also regulates chemicals used during processing and packaging under REACH (Registration, Evaluation, Authorization and Restriction of Chemicals). This covers everything from cleaning agents used on machinery to the materials in your packaging.
Furthermore, general food hygiene standards are non-negotiable. This includes preventing microbial contamination (like Salmonella and E. coli) and mycotoxins (like aflatoxins in nuts and spices), as well as heavy metal contamination (like lead). Acceptable sterilization methods are limited to validated, non-chemical processes like steam sterilization. Gamma irradiation, another common method elsewhere, is highly restricted.
The Zero-Rejection Playbook: Documentation & Logistics
If the three pillars are your strategy, flawless documentation is your execution. EU customs officials are meticulous. Any missing signature, incorrect code, or mismatched detail can hold up your shipment for weeks or lead to outright rejection.
The cornerstone of your documentation package is the electronic Certificate of Inspection (e-COI), which must be created and endorsed in the EU’s TRACES NT (Trade Control and Expert System New Technology) platform before the consignment leaves India. This digital certificate accompanies your shipment, providing a transparent record from origin to destination.
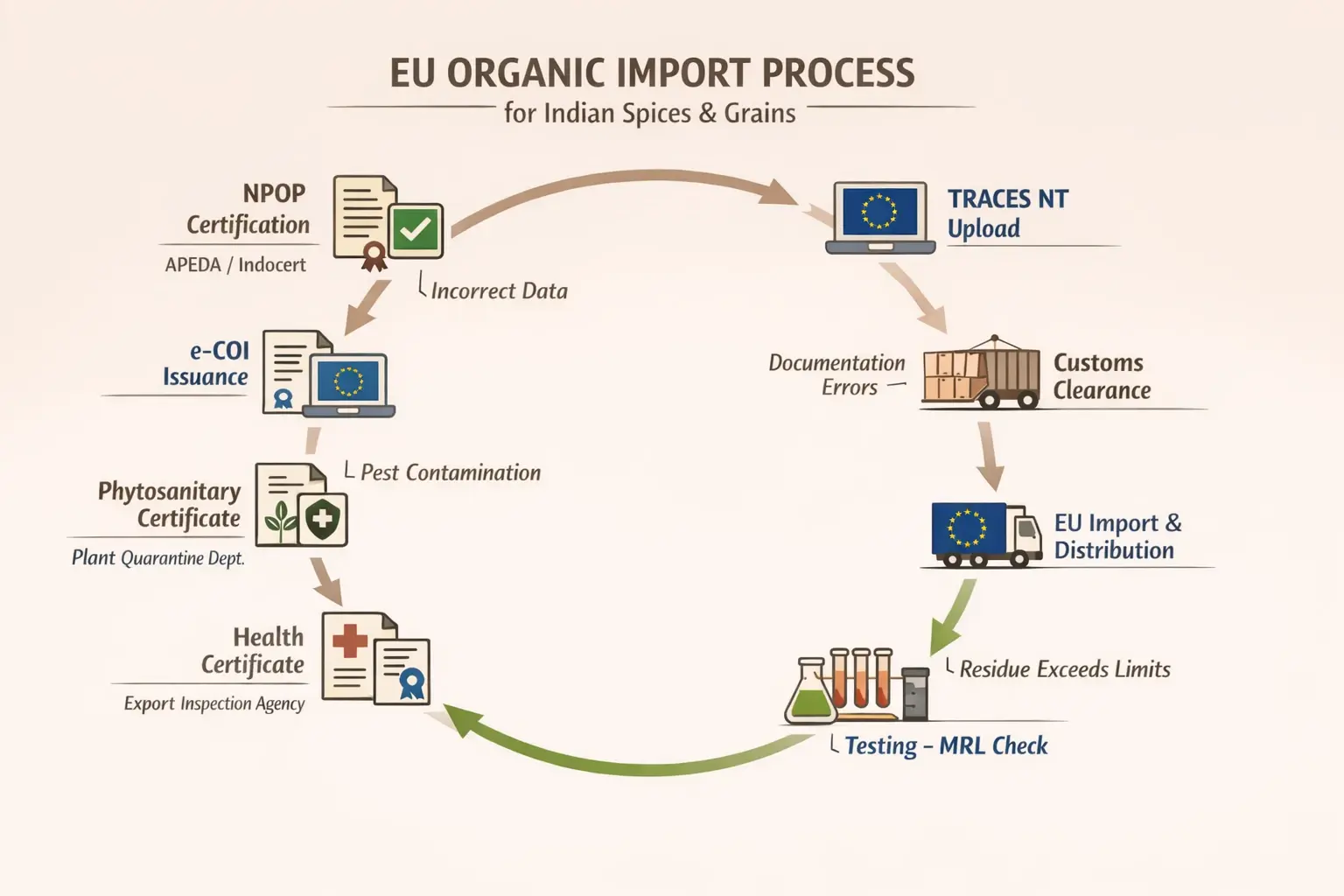
Managing the flow of over a dozen documents—from the phytosanitary certificate to the bill of lading and packing list—requires precision. This is where seamless export logistics become mission-critical. Partnering with experts who understand both Indian export procedures and EU import requirements can prevent simple paperwork errors from becoming million-dollar mistakes.
Finally, physical container integrity is crucial. Proper stuffing, temperature and moisture control, and tamper-proof seals ensure the product that was tested and approved is the exact same product that arrives at the port of Rotterdam, Hamburg, or Felixstowe.

Frequently Asked Questions (FAQ) for EU Organic Imports
What’s the main difference between the old “equivalence” and new “compliance” systems?
Equivalence meant the EU trusted that India’s NPOP standards were “good enough.” Compliance means Indian producers must follow the exact same, highly detailed EU organic regulations as a producer within the EU. The burden of proof is now much higher.
Can I sell my product in the EU if it’s NPOP certified but not specifically for EU compliance?
No. To be sold as “organic” in the EU, the product must be certified by a Control Body recognized by the EU for this specific purpose, and the entire process must follow Regulation (EU) 2018/848. A standard NPOP certification for the domestic market is not sufficient.
What are the most common reasons my organic spice shipment could be rejected?
The top three reasons are: 1) Presence of banned pesticides like Ethylene Oxide (ETO) or residues exceeding MRLs, 2) Incorrect or incomplete documentation, especially issues with the e-COI in the TRACES NT system, and 3) Microbial contamination like Salmonella.
What is TRACES NT, and do I have to use it?
TRACES NT is the EU’s mandatory online platform for managing the sanitary and phytosanitary certification of goods entering the EU. All organic imports require an electronic Certificate of Inspection (e-COI) to be processed through this system before the shipment leaves its country of origin. It is not optional.
How can I find a reliable EU-recognized certification body in India?
The European Commission maintains an official list of recognized Control Bodies in third countries. It is crucial to check this list to ensure the certifier you or your supplier works with is authorized for your specific product category under the new compliance regulation.
Your Path to the EU Market
The EU organic market offers a remarkable premium for those who can navigate its complexities. But success is no longer an accident; it’s a result of a meticulous, proactive strategy.
Achieving zero rejections requires more than just finding a supplier. It demands a holistic approach that integrates deep regulatory knowledge, rigorous custom procurement processes, and end-to-end supply chain control. It means building a resilient system where compliance isn’t just a final check, but a foundational principle from farm to port.
Navigating this process requires deep expertise and on-the-ground presence. Partnering with a specialist who provides end-to-end market-entry solutions can transform this daunting challenge into your company’s most profitable competitive advantage.


