It’s an operations nightmare : a critical shipment of specialty chemicals arrives at your facility from a new supplier. The Certificate of Analysis (COA) looks perfect—all parameters are within spec. But as you integrate the material into your production line, something goes wrong. The yield is off, the reaction is incomplete, or an impurity causes a downstream failure.
The numbers on the COA were correct, but were they reliable? Can we trust these certificates on face value? This question is the silent fear of every procurement leader sourcing from a new market. The answer lies not on the certificate itself, but deep within the supplier’s Quality Management System (QMS)—specifically, in how they validate their test methods and calibrate their equipment.
For international buyers, understanding this process isn’t just a technical detail; it’s the foundation of trust and the key to unlocking the immense potential of the Indian market without the risk. It’s how you move from hoping for quality to ensuring it.

The Bedrock of Quality: Validation, Verification, and Qualification Explained
Before we go deeper, let’s clear up a few terms that are often used interchangeably but mean very different things. Getting this right is the first “aha moment” in understanding your supplier’s true quality maturity.
Think of it like baking a high-stakes, industrial-scale cake:
- Qualification: This is about your tools. Is the oven the right model and installed correctly? (Installation Qualification, IQ). Does it heat to all the specified temperatures accurately? (Operational Qualification, OQ). Does it consistently bake your specific cake recipe to perfection under real-world conditions? (Performance Qualification, PQ). You’re proving the equipment is fit for its purpose.
- Validation: This is about your recipe (the test method). You conduct a series of rigorous experiments to prove that your recipe will consistently produce a perfect cake every single time, even with slight variations in ingredients or environment. Method validation provides documented evidence that a testing procedure is reliable, accurate, and precise for its intended use.
- Verification: This is the final taste test. Before shipping, you bake one more cake using the qualified oven and validated recipe and confirm it meets all quality standards. You’re verifying that a specific batch meets its pre-defined specifications.
Many suppliers focus only on verification—the final COA. But world-class suppliers, and the partners who vet them, know that true quality comes from a deep commitment to qualification and validation. Without them, a good result might just be a matter of luck.
A Look Under the Hood: What is Test Method Validation?
When a supplier validates a test method, they are essentially pressure-testing it from every angle to prove its reliability. For chemical buyers, this is paramount. It ensures that when a COA says a product has 99.8% purity.
Following globally accepted guidelines like ICH Q2(R1), a robust validation process examines several key parameters.
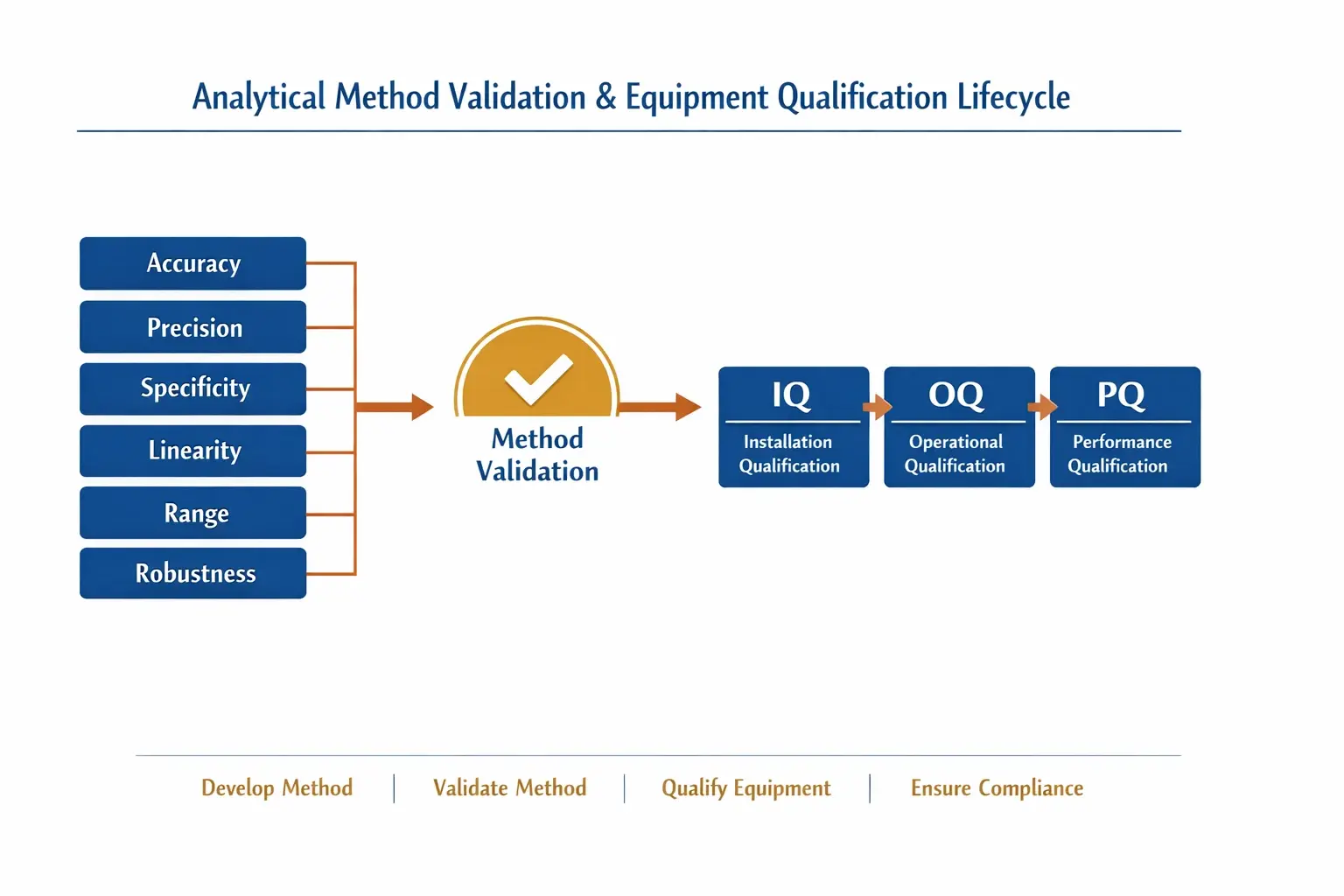
Here’s what your most dependable suppliers are testing for:
- Accuracy: How close is the measured result to the absolute true value?
- Precision: If you run the same test multiple times, how close are the results to each other? This demonstrates repeatability and consistency, ensuring that the first test and the hundredth test are equally reliable.
- Specificity: Does the test measure only the substance it’s supposed to? In a complex chemical mixture, a specific method will accurately measure your target compound without being fooled by impurities or related molecules.
- Linearity & Range: Does the test provide accurate results across a range of concentrations, from low to high? This is crucial for measuring both trace impurities and the purity of the main component.
- Robustness: Can the test method withstand small, deliberate changes in its parameters (like temperature or reagent source) and still produce reliable results?
- Limit of Detection (LOD) & Limit of Quantitation (LOQ): What is the smallest amount of a substance the method can detect versus the smallest amount it can accurately measure? This is critical for controlling harmful or undesirable impurities.
A supplier who can provide a full validation summary report for their test methods is demonstrating a profound commitment to quality that goes far beyond a simple COA. This documentation is a cornerstone of a well-managed QMS and a key indicator of a reliable long-term partner.
The Global Passport: How NABL Accreditation Creates Trust
So, how can you be sure an Indian supplier’s validation and calibration procedures meet global standards without flying across the world to audit them yourself? This is where the Indian regulatory landscape provides a powerful answer: NABL accreditation.
The National Accreditation Board for Testing and Calibration Laboratories (NABL) is India’s national body for accrediting labs. NABL accreditation is not a simple paper certificate; it is a rigorous, on-site verification that a laboratory’s quality management system and technical competence are in line with international standards, primarily ISO/IEC 17025.
Here’s the most important part for international buyers: NABL is a signatory to the Mutual Recognition Arrangement (MRA) of the International Laboratory Accreditation Cooperation (ILAC).
What this means for you: Test results and calibration certificates issued by a NABL-accredited laboratory are officially recognized as equivalent by accreditation bodies in over 100 countries, including the United States, Germany, the UK, and Japan. It is, quite literally, a global passport for quality, eliminating the need for re-testing and building immediate trust in the supplier’s data. When you partner with a supplier whose in-house or third-party lab is NABL-accredited, you are plugging into a system of globally recognized quality.

Common Hurdles and How to Overcome Them
While the framework for world-class quality exists in India, suppliers can face challenges. Legacy equipment may be difficult to qualify, budget constraints can limit investment in new analytical instruments, and skill gaps can make complex validation studies difficult to execute.
This is where having a knowledgeable on-the-ground partner becomes invaluable. A robust sourcing process doesn’t just take a certificate at face value. It involves:
- On-Site Audits: Verifying that the documented procedures for validation and calibration are actually being followed on the laboratory floor.
- Documentation Review: Scrutinizing the supplier’s Validation Master Plan (VMP), individual validation reports, and calibration records to ensure they are thorough and compliant.
- Assessing Third-Party Labs: If a supplier outsources testing, ensuring their chosen lab holds relevant accreditations like NABL is a critical step in the [supplier qualification process].
Navigating these complexities is key to identifying the premier Indian suppliers who are truly ready for the global stage.
Frequently Asked Questions (FAQ)
What’s the main difference between validation and verification again?
Think of it this way: Validation proves the recipe (the method) is good. Verification proves a specific cake (the batch) made with that recipe is good. You validate a method once (with periodic reviews), but you verify every single batch.
Is test method validation a mandatory part of a QMS?
Yes. For any quality system compliant with international standards like ISO 9001 or IATF 16949, ensuring that monitoring and measuring resources are suitable is a core requirement. For labs, ISO/IEC 17025 explicitly requires method validation. It’s a non-negotiable part of a mature QMS.
Why is NABL accreditation better than a standard ISO 9001 certification for a lab?
ISO 9001 certifies that a company has a quality management system in place. However, it does not certify the technical competence of a laboratory to produce reliable test and calibration data. NABL accreditation, based on ISO/IEC 17025, does both. It is the gold standard for proving a lab’s technical proficiency.
What are the first questions I should ask a potential Indian supplier about their testing?
- “Is your in-house laboratory NABL accredited, or do you use a NABL-accredited third-party lab?”
- “Can you provide the validation summary report for the test methods used for our product?”
- “What is the calibration schedule for the key equipment used in this testing, and is it traceable to national standards?”
Their ability to answer these questions confidently and provide documentation is a strong indicator of their quality maturity.
Your Next Step: From Awareness to Action
The numbers on a COA are only the final chapter of a long story. The real story is written in the supplier’s commitment to robust quality systems, proven through rigorous test method validation and diligent equipment calibration.
Understanding these principles empowers you to ask better questions, identify superior suppliers, and build a more resilient and reliable supply chain. It transforms your sourcing strategy from a game of chance into a predictable science.
As you explore [sourcing high-quality chemicals from India], make these technical discussions a central part of your vetting process. By looking beyond the certificate, you will find partners who are not just selling a product, but delivering a promise of unwavering quality.


