A notification arrives. Your shipment from India, perfectly timed to meet production schedules, isn’t on its way to your warehouse. It’s stuck. The U.S. Food and Drug Administration (FDA) has detained it at the port.
Suddenly, your carefully planned supply chain has a critical failure point. Costs mount, production lines are at risk, and you’re facing a complex regulatory challenge you never anticipated.
If this scenario sounds familiar—or it’s the kind of disruption you’re determined to avoid—you’re in the right place. For companies sourcing from India, navigating the FDA’s import process is a mission-critical skill. In fact, data has shown India consistently ranks among the top three countries for food shipment refusals by the FDA, with hundreds of firms on import alert “red lists” for recurring issues.
This isn’t just about avoiding delays; it’s about building a resilient, predictable, and profitable supply chain. Let’s break down why these detentions happen and, more importantly, what you can do about them.
The Basics: What Every Importer Needs to Know
Before diving into prevention, it’s essential to understand the language of FDA imports. These aren’t just bureaucratic terms; they define the challenges you might face.
What is an FDA Detention?
An FDA detention is not a final refusal. It’s an official hold placed on your shipment, indicating that the agency believes the product appears to violate U.S. law. This could be due to anything from suspected contamination to incorrect labeling. During this period, the importer has a window of opportunity—typically 10-20 days—to provide evidence proving the product is compliant.
Think of it as being pulled over for a suspected traffic violation. You haven’t been given a ticket yet, but you need to provide your license and registration to prove you’re compliant.
The “Aha Moment”: Understanding Detention Without Physical Examination (DWPE)
Here’s where many importers get caught off guard. Most assume the FDA physically inspects every container it detains. Often, that’s not the case.
Many detentions happen automatically because of something called Detention Without Physical Examination (DWPE), which is triggered by an Import Alert. If a company, product, or entire country has a history of non-compliance for a specific issue (like Salmonella in spices), the FDA can place them on an Import Alert “Red List.”
This means all future shipments of that product from that supplier will be automatically detained upon arrival. No inspection is needed to trigger the hold. The burden of proof is immediately shifted to the importer to prove the specific shipment is safe and compliant before it can be released.
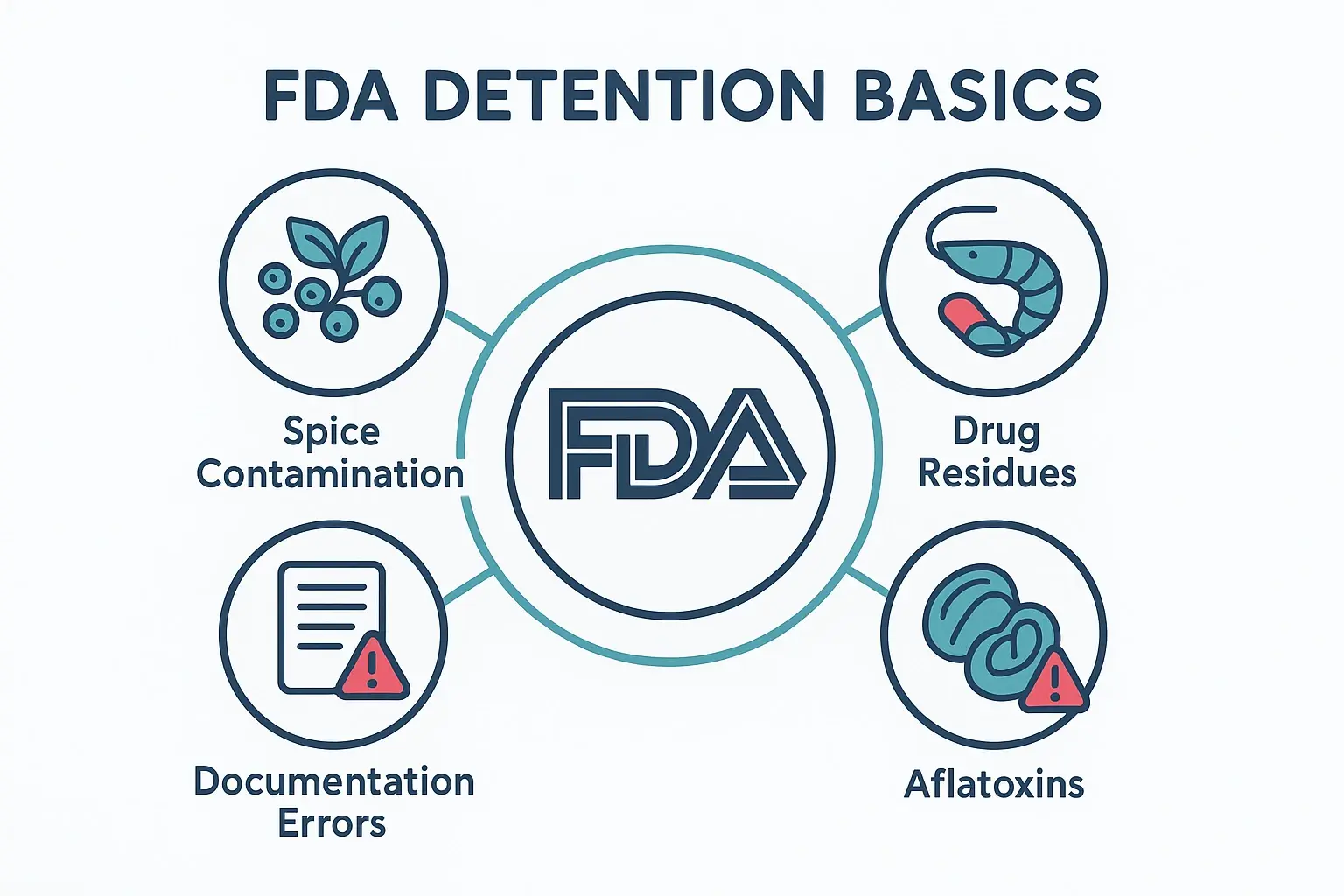 This visual summarizes core FDA detention reasons for Indian food shipments, highlighting major product issues and regulatory categories to build foundational understanding.
This visual summarizes core FDA detention reasons for Indian food shipments, highlighting major product issues and regulatory categories to build foundational understanding.
The Root of the Problem: Top Reasons Indian Food Shipments Are Flagged
While reasons for detention can be vast, a few common themes emerge for products sourced from India. They fall into two main categories: issues with the product itself and errors in the paperwork.
Product-Specific Violations: The Usual Suspects
- Microbiological Contamination: This is a major one, especially for spices. Salmonella contamination in black pepper, for example, is a well-documented issue that has led to multiple firms being placed on Import Alert. Around 70% of the nearly 200 Indian firms on the red list for Salmonella are spice companies.
- Veterinary Drug Residues: The Indian seafood industry, particularly farmed shrimp, has faced scrutiny for the presence of unapproved antibiotic residues. The FDA has a zero-tolerance policy for these substances, leading to automatic DWPE for listed shippers.
- Filth and Foreign Matter: This charge means the product contains things that shouldn’t be there, like mold, insects, or other adulterants. Historically, this has been an issue for products like black pepper and processed goods like papad.
- Pesticides and Aflatoxins: Agricultural products like tree nuts, dried fruits, and seeds are frequently tested for illegal pesticide residues and aflatoxin (a toxic mold byproduct). Shipments exceeding the FDA’s strict limits are promptly detained.
The Paperwork Paradox: How Minor Clerical Errors Cause Major Delays
Even if your product is perfect, your shipment can still be detained. Experts at organizations like Certified Laboratories point out that simple, avoidable administrative mistakes are a leading cause of delays.
The top five entry submission errors include:
- Missing Affirmation of Compliance (AofC) Codes: These are mandatory codes that affirm the product complies with specific FDA requirements.
- Incorrect Manufacturer Information: Using the headquarters address instead of the actual manufacturing site is a common mistake. The FDA’s Facility Establishment Identifier (FEI) number must point to the physical location where the food was produced.
- Wrong Product Codes: The FDA uses a highly specific coding system to classify products. A small error can lead to a mismatch and trigger a detention for clarification.
- Inaccurate Quantity and Packaging Information: The numbers on the entry documents must exactly match the physical shipment.
- Wrong Shipper or Consignee: A simple typo in a company name or address can cause the system to flag the entry.
These may seem like minor details, but to the FDA’s automated screening system (PREDICT), they are red flags that signal a potential problem, forcing a manual review and an immediate delay.
The Shift to Prevention: Building a Detention-Proof Supply Chain
Reacting to a detention notice is stressful and expensive. The real solution lies in a proactive approach that addresses risks long before your container is loaded.
It Starts at the Source: Proactive Supplier Vetting
You cannot afford to be surprised. The single most effective way to prevent detentions is to work with suppliers who have a proven track record of FDA compliance. A partner with on-the-ground presence can perform the necessary diligence. This involves a (supplier qualification) process that verifies:
- FDA Registration: Is the facility properly registered?
- Compliance History: Are they currently on an FDA Import Alert list?
- Quality Control: Do they have robust testing protocols for common contaminants like Salmonella or pesticides?
- FSMA Compliance: Do they have a Foreign Supplier Verification Program (FSVP) plan in place, as required by the Food Safety Modernization Act?
Documentation Perfection: Your First Line of Defense
Treat your shipping documents with the same care as the product itself. Before any shipment, create a pre-flight checklist to ensure every detail is perfect—from the FEI number to the product codes and packaging counts. This simple step can eliminate the majority of administrative detentions.
 Depicts the top FDA entry submission errors hindering Indian food shipments and the sequential prevention steps exporters should follow.
Depicts the top FDA entry submission errors hindering Indian food shipments and the sequential prevention steps exporters should follow.
Crisis Management: You’ve Received a Detention Notice. Now What?
Even with the best preparation, detentions can happen. When they do, clear, decisive action is crucial.
Your Golden Window: Responding Effectively
As legal experts at Garg Law emphasize, the period after receiving a Notice of FDA Action is your “most important” opportunity to save the shipment. You have a limited time to present evidence that your product is, in fact, compliant.
This is not the time for a simple email. A strong response includes:
- A clear, written argument addressing the specific violation cited.
- Third-party laboratory analysis of the detained product.
- Supporting documentation about the manufacturer’s quality control processes.
Reconditioning: Can Your Shipment Be Saved?
In some cases, the FDA may permit the importer to “recondition” the product to bring it into compliance. This could involve relabeling packages to correct a misbranding charge or treating a product to eliminate microbiological contamination.
This process requires a formal proposal to the FDA and must be performed under their supervision. While not always possible, it can be a viable alternative to having the shipment destroyed or re-exported, saving significant costs. Navigating the complexities of (export logistics) for re-export can often be more costly than reconditioning.
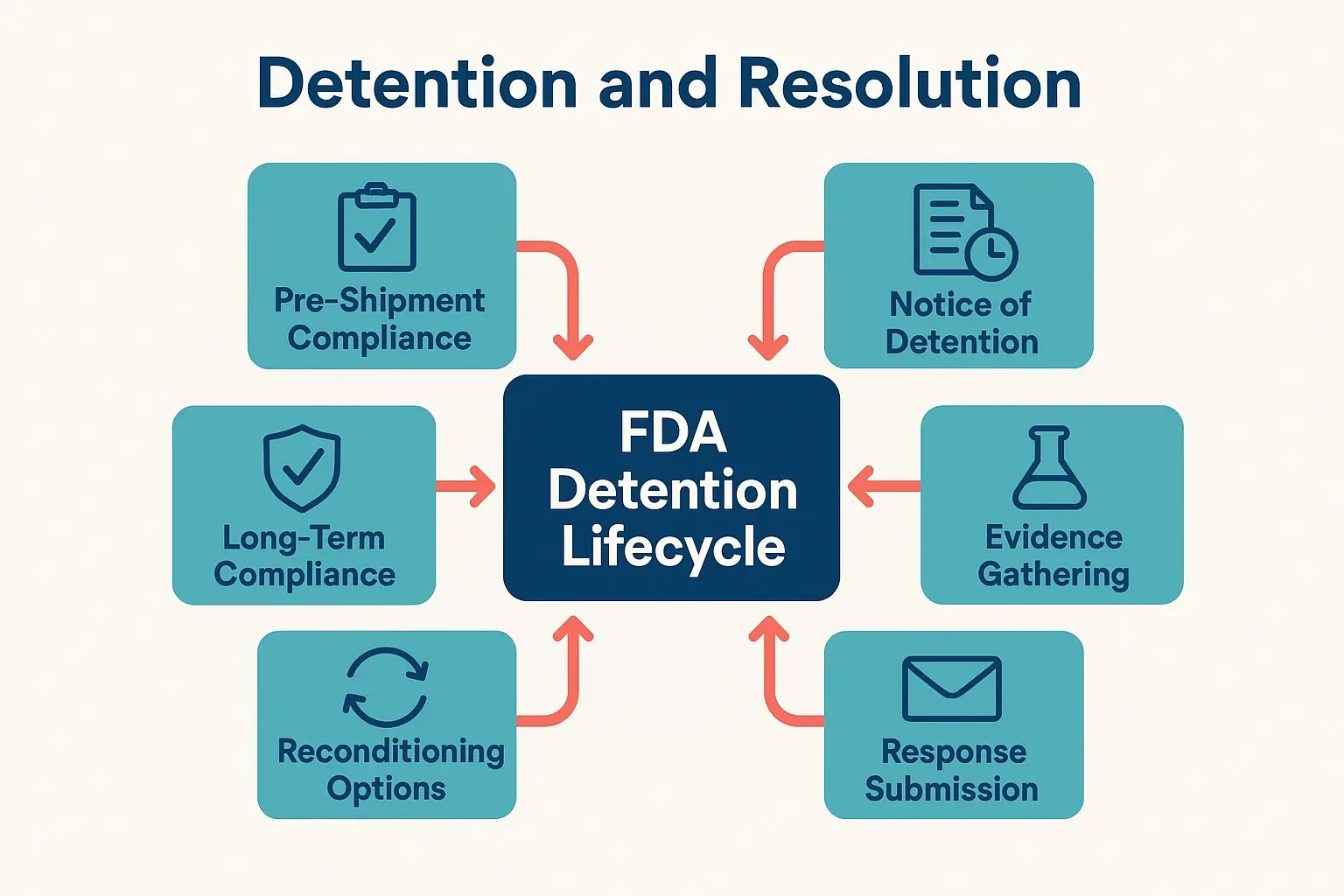 Visualizes the comprehensive detention lifecycle and resolution process including pre-shipment compliance, notice response, reconditioning, and long-term strategies.
Visualizes the comprehensive detention lifecycle and resolution process including pre-shipment compliance, notice response, reconditioning, and long-term strategies.
The Long Game: Getting Off the Red List and Staying Compliant
The ultimate goal is not just to resolve a single detention, but to build a reputation for compliance that keeps you off the FDA’s radar. For firms on a DWPE “Red List,” the path to redemption involves petitioning the FDA for removal.
This requires submitting a comprehensive package demonstrating that the root cause of the problem has been fixed. Typically, this includes evidence of at least five consecutive compliant shipments, verified by a third-party lab. Achieving this “Green List” status exempts the firm from automatic detention and is the gold standard for any exporter. This is where a partner providing (custom procurement) solutions can manage the entire compliance journey.
Frequently Asked Questions About FDA Detentions
What’s the difference between an FDA hold and a Customs hold?
An FDA hold relates to the product’s compliance with food, drug, and cosmetic safety laws. A Customs and Border Protection (CBP) hold is typically related to import duties, tariffs, intellectual property, or security concerns. A shipment can be held by both agencies.
How long can my shipment be detained?
The initial detention period gives the importer a chance to respond. If a response is filed, the process can take several weeks or even months as the FDA reviews the evidence. If no response is filed, the shipment will be refused entry.
Is it true that certain Indian certifications can help with FDA compliance?
While certifications from bodies like India’s Export Inspection Council (EIC) are valuable indicators of quality, they do not grant automatic exemption from FDA regulations. The FDA operates solely under U.S. law. However, a supplier with strong local certifications is often more likely to meet FDA standards.
Your Partner in Compliance and Growth
Successfully importing food products from India to the U.S. is less about luck and more about a disciplined, proactive strategy. It requires a deep understanding of regulations, rigorous (boots-on-the-ground supplier audits), and flawless execution of logistics and documentation.
By understanding the common pitfalls and building a supply chain based on prevention, you can turn a potential liability into a competitive advantage, ensuring your products flow smoothly from port to customer without costly interruptions.