An urgent notification pops up. A critical batch of specialty chemicals from your new supplier in India is showing a slight color deviation on the Certificate of Analysis (COA). It’s 10 PM your time, morning in Gujarat. What follows is a frantic scramble of emails with low-resolution photos, time-zone-delayed conference calls, and a growing sense of uncertainty. Is this a minor issue or a deviation that could shut down your production line?
For any global procurement manager sourcing from India, this scenario is a familiar source of anxiety. The distance isn’t just geographical; it’s an information gap where quality concerns can multiply. But what if you could close that gap? What if you could have a real-time, transparent view into your supplier’s quality process, turning reactive firefighting into proactive management?
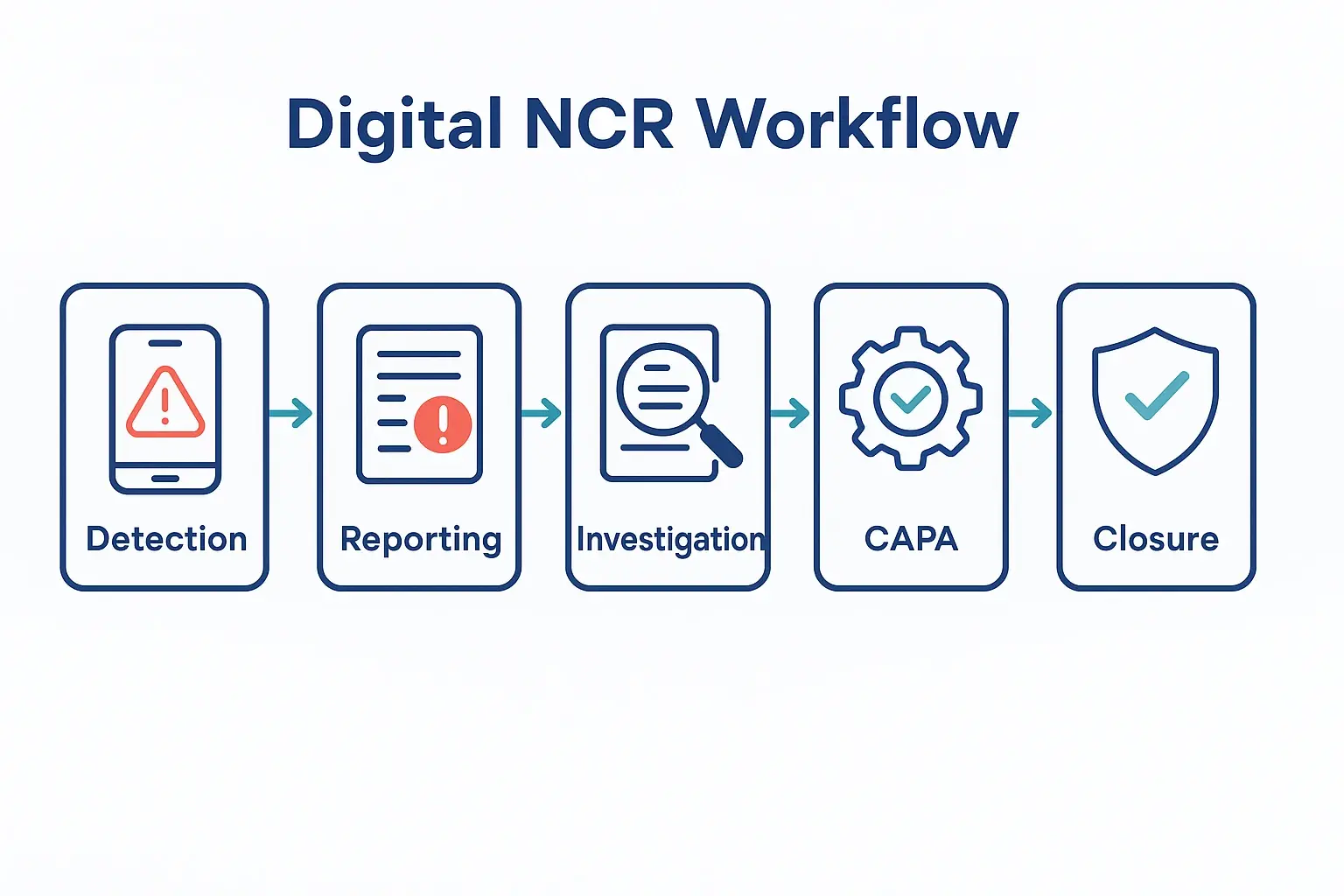
This is the power of a well-implemented digital Non-Conformance Reporting (NCR) system. It’s not just another piece of software; it’s a framework for trust, transparency, and control across continents.

First, Let’s Clear the Air: What is an NCR in Quality Management?
Before we dive in, let’s clarify a common point of confusion. If you search for “NCR system,” you’ll likely see results for NCR Voyix, a company specializing in point-of-sale and retail technology. That’s not what we’re talking about.
In the world of manufacturing and supply chain, an NCR stands for Non-Conformance Report. It’s a formal, documented flag raised whenever a product, process, or material fails to meet a specified requirement. Think of it as the foundational document for quality control.
An NCR isn’t just about pointing out a problem. As defined by quality standards like ISO 9001, it’s the official starting point for:
- Containment: Immediately isolating the affected product.
- Investigation: Performing a Root Cause Analysis (RCA) to understand why it happened.
- Disposition: Deciding what to do with the non-conforming product (rework, scrap, use-as-is).
- Correction: Implementing Corrective and Preventive Actions (CAPAs) to ensure it never happens again.
This process is a cornerstone of any robust Supplier Quality Management (SQM) system, which is the overall strategy for monitoring and improving the performance of your suppliers.
Why Traditional Quality Control Falls Short Across Continents
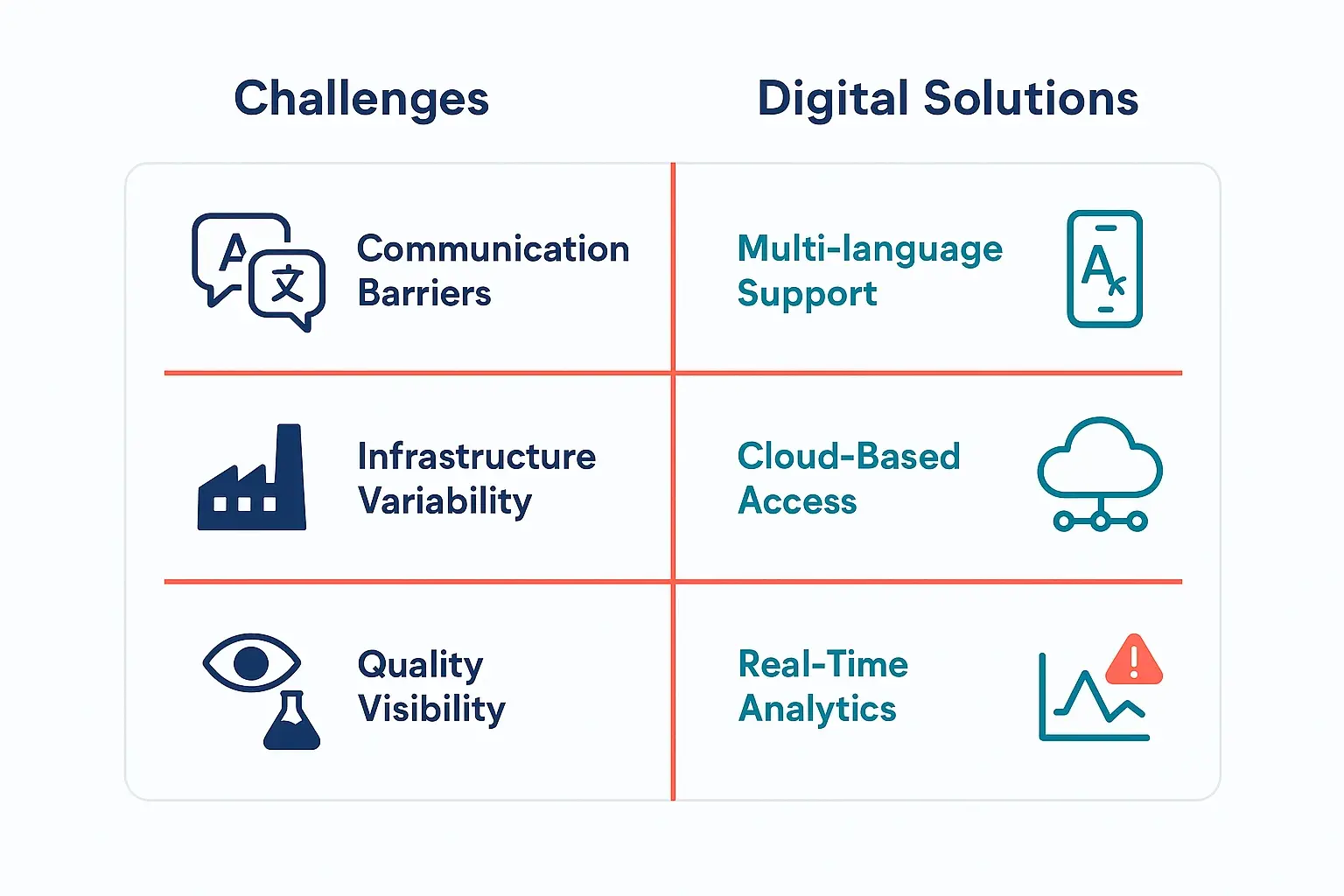
For decades, NCRs were managed with paper forms, spreadsheets, and endless email chains. This might work when your supplier is across town, but it creates critical vulnerabilities when managing partners thousands of miles away in a different time zone.
The traditional approach leads to several challenges that global chemical buyers know all too well:
- Lack of Visibility: A quality issue might not be discovered until the shipment arrives at your port, weeks after it was produced. By then, it’s too late.
- Delayed Communication: Time zone differences and language barriers can turn a simple clarification into a multi-day email exchange, delaying critical decisions.
- Fragmented Data: Evidence—like photos, batch records, and lab tests—is often scattered across different inboxes and folders, making a comprehensive investigation nearly impossible.
- Reactive Problem-Solving: Without real-time data, you’re always playing catch-up, addressing problems after they’ve already impacted your schedule and budget.
These issues are magnified in the chemical industry, where purity, consistency, and compliance aren’t just quality metrics—they’re matters of safety and regulatory necessity. A minor deviation can have major consequences.
The Digital NCR Blueprint: A Step-by-Step Guide for Remote Management
Establishing a digital NCR system transforms this dynamic. It creates a single source of truth that is accessible to both you and your Indian supplier 24/7, fostering a partnership built on shared data and accountability. Here’s how to build it.
Phase 1: Laying the Foundation
Success begins long before the first non-conformance is reported. This phase is about setting clear, mutually understood expectations.
- Define Unambiguous Quality Standards: You can’t report a deviation if the standard isn’t clear. Ensure your technical specifications, COA requirements, Safety Data Sheets (SDS), and packaging standards are documented and shared. This becomes the “constitution” for your quality agreement.
- Select the Right Digital Platform: Not all systems are created equal. For remote management, prioritize platforms with:
- Cloud-Based Accessibility: Accessible from any device, anywhere.
- Mobile-First Design: Enables your supplier’s on-site QC team to report issues directly from the plant floor with a phone or tablet.
- Multimedia Uploads: The ability to attach high-resolution photos, videos, and documents is non-negotiable for remote validation.
- Automated Workflows & Alerts: Instantly notifies the right people (on both sides) when an NCR is raised or updated.
- Onboard and Train Your Indian Supplier: Technology is only a tool; people make it work. A crucial step in any successful supplier qualification process is collaborative onboarding. Conduct joint training sessions (even virtually) to demonstrate the system, explain its benefits for them (e.g., faster resolution, clearer communication), and establish clear protocols for its use.
Phase 2: Mastering Remote Operations
With the foundation in place, the system becomes your eyes and ears on the ground.
- Real-Time Reporting: An operator at your Indian supplier’s facility notices a slight pH variance in a pre-shipment sample. Instead of waiting for a shift report, they use their mobile device to open a new NCR, upload a photo of the lab reading, and input the batch number—all in under five minutes. You receive an instant alert.
- Collaborative Investigation: The digital NCR form guides the supplier through the initial investigation. You can see their findings and collaborate on a Root Cause Analysis using integrated tools or by referencing the shared data on a video call. The entire history is captured in one place.
- Trackable CAPAs: Once a corrective action is agreed upon (e.g., “re-calibrate pH meter and retrain operator”), it’s logged in the system. You can assign owners, set deadlines, and require verification evidence (like a photo of the new calibration certificate) before the NCR is closed. This ensures that fixes are implemented, not just discussed.
Navigating the Nuances: Digital NCR in the Indian Chemical Ecosystem
Implementing any system requires an understanding of the local context. A digital NCR framework is uniquely suited to address the specific challenges of remote management in India and turn them into strengths.
For instance, while verbal agreements and relationship-based business are common, a digital system introduces a layer of structured clarity that benefits both buyer and supplier. It ensures that critical quality details are not lost in translation or informal communication. An effective compliance support strategy relies on this kind of documented precision.
By providing a clear, simple, and mobile-friendly tool, you can also overcome potential infrastructure variability. The system ensures that reporting remains consistent and accessible, regardless of location.
Beyond Firefighting: Using NCR Data for Proactive Improvement
The true value of a digital NCR system emerges over time. It’s not just about solving individual problems; it’s about preventing future ones.
With a few clicks, you can access dashboards and run reports to identify trends. You might discover that:
- A specific product line accounts for 60% of all non-conformances.
- Deviations spike during a particular production shift.
- A certain raw material supplier is consistently linked to batch impurities.
This data is gold. It allows you to move from being a reactive buyer to a strategic partner, working with your supplier on targeted continuous improvement initiatives. This is the goal of a true end-to-end supply-chain management partnership: using data to make the entire chain stronger, more resilient, and more predictable.
Frequently Asked Questions (FAQ)
What’s the difference between a major and minor non-conformance?
A minor non-conformance is a small deviation that doesn’t affect the product’s fit, form, or function (e.g., a slight labeling error). A major non-conformance is a significant failure to meet requirements that could impact performance, safety, or regulatory compliance (e.g., incorrect chemical purity). Your digital system should allow for this classification.
Who is responsible for issuing an NCR?
Typically, anyone involved in the quality process can raise an NCR—from a QC technician on the supplier’s floor to your own team during an incoming inspection. A good digital system will have defined roles and permissions for raising, reviewing, and approving NCRs.
How does a digital NCR system integrate with our existing ERP?
Many modern QMS and NCR platforms offer APIs (Application Programming Interfaces) that allow them to connect with Enterprise Resource Planning (ERP) systems. This can automate processes like putting non-conforming inventory on hold or linking quality data to specific purchase orders.
What is the typical technology readiness of Indian chemical suppliers?
This varies widely, but it’s a mistake to assume a lack of readiness. Many leading Indian manufacturers are technologically sophisticated and already use digital quality systems. For others, the key is to choose a system that is highly intuitive, mobile-friendly, and requires minimal IT infrastructure. The focus should be on simplicity and a clear value proposition for the supplier.
Your Roadmap to Remote Quality Excellence
Shifting from spreadsheets and emails to a digital NCR system is a powerful step towards building a more resilient and transparent global supply chain. It replaces ambiguity with data, distance with visibility, and reactivity with proactivity.
By creating a shared framework for quality, you’re not just buying chemicals; you’re building a partnership with your Indian supplier founded on trust and a mutual commitment to excellence. You’re bridging the distance, one data point at a time.
Looking to seamlessly procure goods from India to the USA? Get in touch with our team today!