You’ve done the work. You identified a promising chemical manufacturer in India, vetted their capabilities, and approved the sample. The first production batch arrives, and the Certificate of Analysis (COA) matches the specifications perfectly. But when your team uses the material in a formulation, something is off. The reaction yield is low, the color is wrong, or an unexpected side product appears.
The COA said it was fine. So, what went wrong?
This scenario is a common frustration for procurement managers sourcing chemicals globally. It’s a classic case where the numbers on a page don’t tell the whole story. The real issue isn’t just a deviation; it’s a hidden root cause that a standard quality check might miss. To solve it, you need to go beyond basic problem-solving and become a chemical detective. This requires a specialized approach to Root Cause Analysis (RCA) tailored for the unique complexities of chemical manufacturing in India.
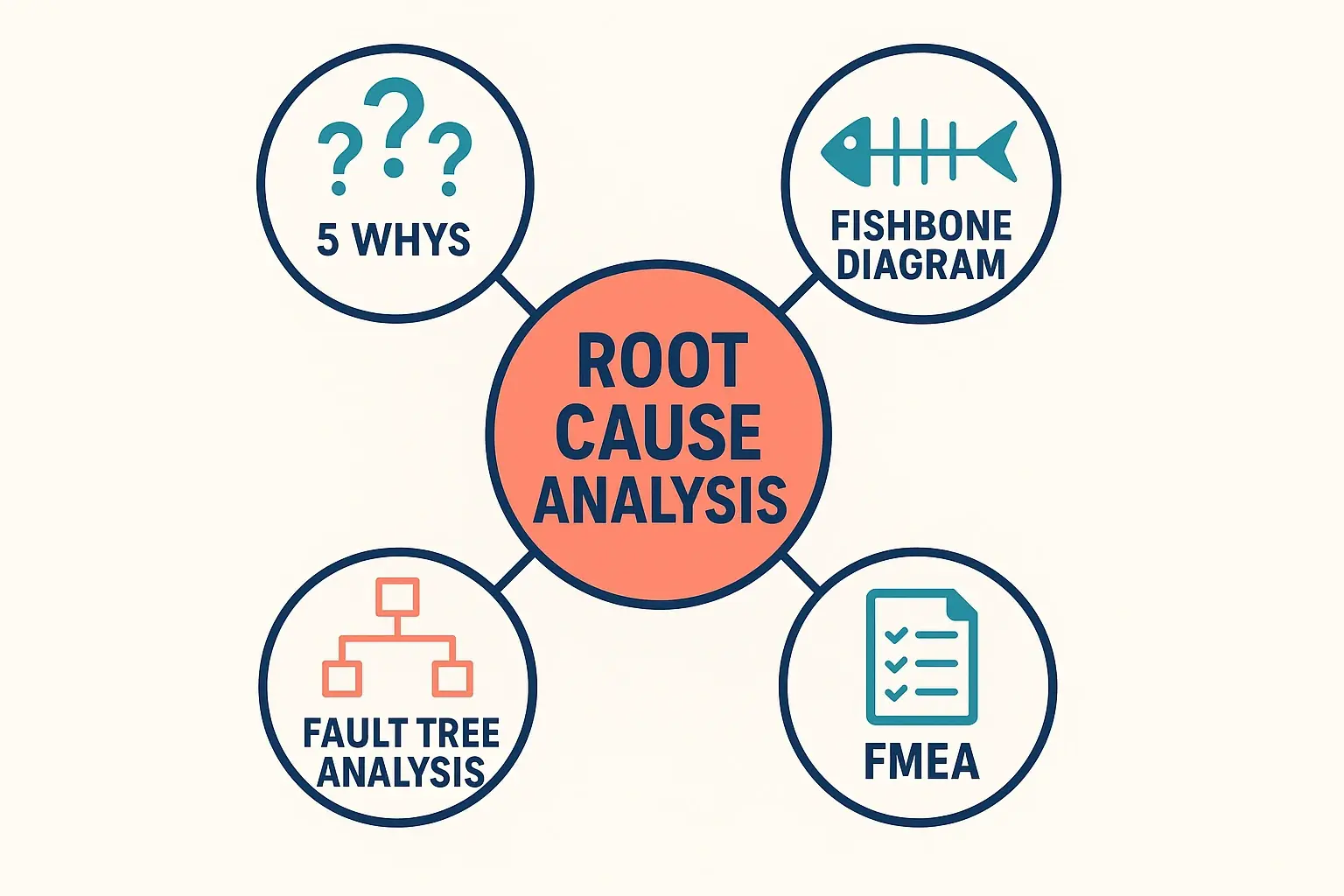
Why Standard RCA Often Falls Short for Chemical Purity
Root Cause Analysis is the process of digging deeper than the surface-level problem to find the fundamental cause. Most of us are familiar with basic RCA tools like the “5 Whys”—simply asking “Why?” repeatedly until you land on the core issue.
While helpful, this approach can be too simplistic for chemical purity deviations. The problem is rarely a single, linear failure. A trace impurity at 0.1% could be caused by a complex web of factors, including:
- Complex Reaction Kinetics: A slight temperature fluctuation changing a reaction pathway.
- Trace Contaminants: A previously undetected heavy metal in a raw material acting as an unwanted catalyst.
- Analytical Blind Spots: The testing method used for the COA isn’t designed to detect the specific impurity causing the problem.
- Environmental Factors: High humidity during a drying phase leading to excess residual moisture.
Simply asking “Why?” five times won’t uncover a subtle shift in a chromatographic baseline. To truly succeed, you need to adapt classic RCA frameworks to the realities of the lab and the production floor.
Tailoring RCA Tools for the Chemical Lab
Instead of abandoning proven RCA tools, the key is to enrich them with chemical and process engineering knowledge. Think of it as adding high-resolution lenses to your investigative toolkit.
The Fishbone Diagram: Deconstructing a Purity Problem
The Fishbone (or Ishikawa) Diagram is perfect for brainstorming all the potential causes of a complex problem. Let’s imagine a common scenario: “High Level of Impurity X in Final Product.”
A tailored Fishbone Diagram would explore specific chemical-related causes:
- Raw Materials:
- Was there variability in the quality from the Indian raw material supplier?
- Did a new, unvetted secondary supplier get used?
- Could contamination have occurred during transport or storage? A robust supplier qualification process is your first line of defense here.
- Process Parameters:
- Was the reaction temperature or pressure outside the validated range?
- Was the mixing time or speed incorrect, leading to an incomplete reaction?
- Was the pH not properly controlled at a critical step?
- Equipment:
- Could there be residue or cross-contamination from a previous batch in the reactor?
- Is there corrosion on a valve or pipe leaching metals into the product?
- Was a filter compromised, allowing particulates through?
- Analytical Methods:
- Is the testing method (e.g., HPLC, GC) sensitive enough to detect the impurity at the required level?
- Was the equipment calibrated correctly?
- Could the sample itself have been contaminated during collection or preparation?
- Personnel & Environment:
- Was there a deviation from the Standard Operating Procedure (SOP)?
- Are operators fully trained on the subtleties of the process?
- Could high ambient humidity or temperature have affected a sensitive step?
This detailed approach transforms a generic problem-solving tool into a powerful diagnostic map specific to your chemical process.
The “India Factor”: Unique Challenges in Sourcing and RCA
Investigating a quality deviation requires understanding the specific operational context. Sourcing from India offers immense advantages, but it also comes with unique variables that must be factored into any root cause analysis.
Supply Chain and Raw Material Variability
Many Indian manufacturers source their own raw materials from a diverse network of local suppliers. This can introduce variability that impacts final product consistency. A thorough RCA must investigate the entire upstream supply chain. Were the incoming raw materials properly tested? Was there a change in the raw material supplier that wasn’t communicated? This is a critical area where having on-the-ground support is invaluable for navigating chemical sourcing from India effectively.
Environmental and Infrastructural Nuances
Factors we often take for granted can play a significant role. Seasonal changes, like the monsoon season, can dramatically increase humidity, affecting drying processes or causing moisture-sensitive materials to degrade. Power grid fluctuations can impact the stability of sensitive electronic equipment controlling a reaction. A good RCA considers these external variables, not just what happens inside the reactor.
Your “Chemical Detective” Toolkit: Going Beyond the Obvious
A successful investigation hinges on asking the right questions and knowing where to look for clues. The COA is just the starting point; the real evidence is often hidden in the analytical data and the process itself.
Reading the Clues in the Data
Don’t just look at the final purity percentage. Look at the raw data from analytical tests like High-Performance Liquid Chromatography (HPLC) or Gas Chromatography (GC).
- Look for small, unidentified peaks: An unexpected “blip” on a chromatogram, even if small, could be the impurity causing your issue.
- Analyze peak shape: Poorly shaped peaks can indicate problems with the analytical method or co-elution with another substance.
- Compare batch-to-batch data: Overlaying the analytical data from a “good” batch and a “bad” batch is one of the fastest ways to spot differences.
Asking the Right Analytical Questions
When an unknown impurity appears, frame your investigation with a checklist:
- Is it related to a starting material or reagent?
- Is it a known by-product of a side reaction?
- Is it a degradation product of the final molecule?
- Is it contamination from an external source (e.g., solvent, catalyst, equipment)?
Each question points to a different branch of the Fishbone Diagram and helps focus your investigation. Success requires a partnership where your supplier is willing and able to help you run these analytical diagnostics—a key component of a comprehensive quality assurance program.
Frequently Asked Questions (FAQ) about RCA for Chemical Purity
What’s the first step when a chemical batch is out of specification?
First, quarantine the batch to prevent its use. Then, immediately communicate with your supplier, providing all relevant data—the COA, your internal test results, and a clear description of the problem observed in your application. A transparent, data-driven conversation is key.
Should every minor deviation go through a full RCA?
Not necessarily. It’s about risk. A minor color deviation in an industrial solvent might be acceptable, while a 0.1% unknown impurity in a pharmaceutical intermediate requires a full, rigorous investigation. Use a risk-based approach to decide how deep to go.
How can I prevent these deviations in the first place?
Prevention is always better than correction. It starts with a rigorous supplier qualification, clear and detailed specification sheets, and a robust quality agreement. It also involves working with partners who have strong on-the-ground process controls and a culture of proactive quality management.
What’s the difference between a Corrective Action and a Preventive Action (CAPA)?
A Corrective Action fixes the immediate problem (e.g., “re-purify the off-spec batch”). A Preventive Action addresses the root cause to ensure the problem never happens again (e.g., “install a new temperature sensor and alarm on the reactor to prevent overheating”). A true solution always includes a preventive action.
Building a Resilient Chemical Supply Chain
Dealing with chemical purity deviations isn’t just about fixing a bad batch; it’s about building a more resilient and predictable supply chain. Effective Root Cause Analysis is a critical skill, but it shouldn’t be a daily fire drill.
The ultimate goal is to work with sourcing partners who have this “chemical detective” mindset built into their DNA. A partner who understands the nuances of both Western quality expectations and Indian manufacturing realities can help you move from reactive problem-solving to proactive prevention.
By mastering the principles of specialized RCA, you can transform quality challenges from frustrating roadblocks into opportunities for process improvement, strengthening your products and your supplier relationships for the long term.